Original paper: Direct observation of DNA knots using a solid-state nanopore
Try taking out your earphones from your pocket and, in all probability, you’ll find knots and entanglements between the ends. As it turns out, this knotting effect is not limited to macroscopic objects, but occurs on the nanoscale as well. A DNA molecule that carries the genetic information of a living organism is actually a long string-like polymer, so you can imagine that it would also get tangled up just like the cords of your earphones. In fact, scientists know that DNA does form knots when it is in the nucleus of a cell, and these knots need to be removed by specialized bio-molecules, called enzymes, so that a cell can ‘read’ the genetic information encoded in the DNA. [1] In today’s paper, Calin Plesa and his colleagues at TU Delft are able to observe and measure these knots in DNA strands. In the process, they also observe interesting knotting behaviour which has not been observed before.
Knots on DNA

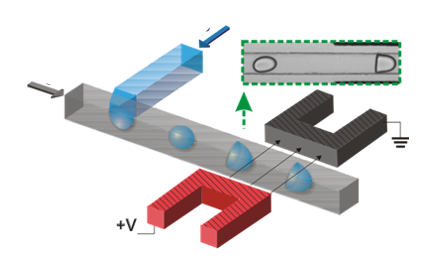
The researchers use a nanopore sensor to infer the structural properties of a DNA molecule. The sensor is made up of two reservoirs filled with electrolyte (a solution which separates into cations and anions, which can be used to conduct electricity, e.g. a salt solution), and they are separated by a membrane, or thin sheet, with a tiny hole in it. An electric field applied across the membrane generates an ionic current in the electrolyte and also pulls a negatively charged DNA strand through the tiny opening. The passage of a DNA strand through the nanopore causes a dip in the ionic current that is proportional to the volume of ions displaced—in other words, it’s proportional to the size of the molecule (a typical scenario is shown in Figure 1). Therefore, a knot in the DNA can generate a bigger drop in the current than an untangled strand. From this difference it is possible to infer the characteristics of the knot itself, since a bigger drop indicates a bigger knot.
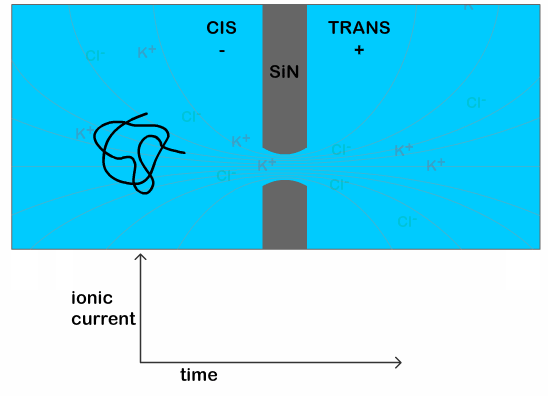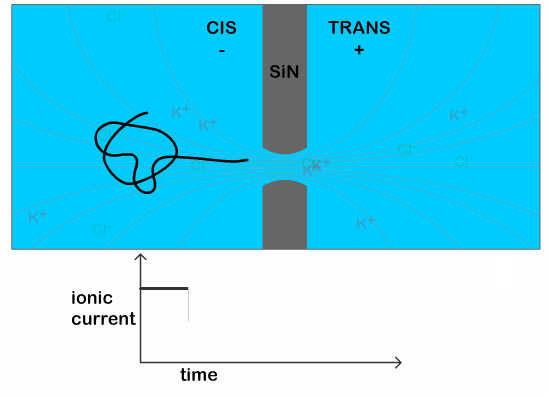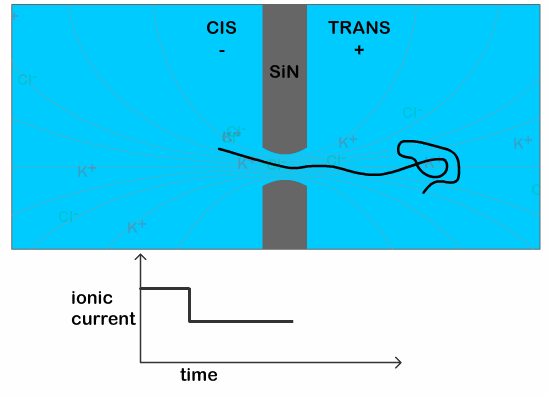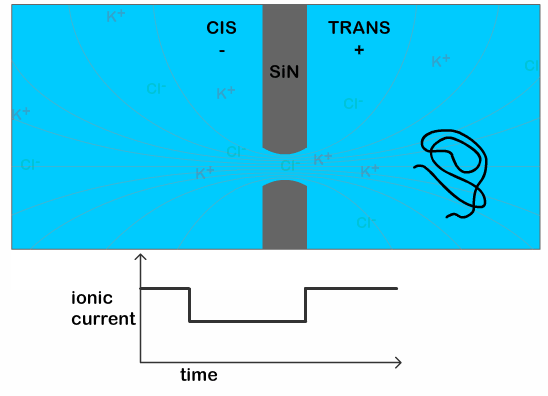
The typical time for a DNA to pass through the pore is in the order of a few milliseconds, when the DNA is in a solution of potassium chloride (which is the typical salt solution used to carry out nanopore experiments). This makes it difficult technically, to see any features present on the DNA. Previous work has shown that it is possible to slow down the DNA passage by at least 10 times by using lithium chloride as their salt solution. [3] This increase in the translocation time (time it takes for the DNA to pass through the pore) is necessary to clearly see the additional dip in the current as the knot traverses the pore, as illustrated in Figure 2.

The dip in the current signal caused by the knot passing through the pore can then be used to infer characteristics about the knot. In particular, it can be used to calculate the size of the knot, which has not been experimentally determined before. This has both physical and biological significance. Physically, it helps us understand the types of knots being formed on polymers as it can tell us whether the knot is loose or tightly formed. Biologically, it can help us understand how naturally occurring enzymes are able to disentangle knots in DNA strands, a function which is still poorly understood. The size of the knot is estimated by using
$latex d= v t$
where d is the length of the knot along the DNA strand, v is the average speed of the DNA translocation, and t is the time the knot takes to traverse the pore. Using this technique, the researchers estimate that the majority of the knots are less than 100 nm long. Previous research has shown that the DNA strand is rigid over lengths shorter than 50 nm, so considering this, the estimated knot size suggests that the knot is very tight. [2] However, this result needs further analysis, as the process of pulling the DNA through the nanopore might cause the knot to tighten, so this might not be the knot’s size in its natural state.
Slipping and sliding knots
When considering a linear (think: a thread with loose ends) DNA molecule, there is a possibility of the knot ‘slipping’ off the end of the strand before it gets pulled into the nanopore. For the knot to traverse the pore, it needs to be pulled fast enough to get squeezed to the size of the pore. If this process doesn’t happen fast enough the knot ‘halts’ at the pore entrance while the unknotted region translocates through. This allows the knot to disentangle, in case of a linear DNA molecule.
To determine if this slipping process occurs in knotted DNA strands, the researchers repeat their experiment using a circular (think: a thread joined end-to-end) DNA molecule. By using a closed loop they avoid possibility of the knot disentangling, but the knot can still slip towards the trailing end of the DNA during the translocation. The position of the knot is determined by the position of the dip in the current signal (purple line in Figure 2). They measure the probability of finding the knot at each position along the strand using two voltages, 100 mV and 200 mV. As shown in Figure 3, the knots show a preference for sliding toward the trailing end of the molecule at higher voltages, indicating that pulling too hard on the leading end of the DNA strand can indeed cause knots to slip along the strand instead of being pulled through the pore. The researchers also observe a 55% higher knotting occurrence in the circular molecules compared to linear ones. This suggests that knots may have slipped off the end of the linear molecules, thereby not detecting them at all.

The researchers in this study have shown that naturally induced knots occur in DNA strands and they measured the sizes of those knots, which were previously unknown. This measurement showed that the knots detected are actually quite tight, which was not expected, although this result still needs to be investigated further. Additionally, these knots were seen to slide along the DNA molecules as they traversed the nanopore due to the strong pull at the end of the DNA strand. This was seen clearly by repeating the knotting experiments using circular DNA where there were no ends for the knots to slide off.
This new information about the structure of knots in DNA strands will help inform future studies of the complex topological structures formed in biomolecules such as DNA and proteins. It will also contribute to understanding the effects of topological features on the biological functions of these long, string-like biomolecules. In effect, it can help us explain the consequence of knotted DNA on the cell’s function as well as how the cell is equipped to handle these defects.
[1] http://www.tiem.utk.edu/~gross/bioed/webmodules/DNAknot.html
[2] Baumann, Christoph G., et al. “Ionic effects on the elasticity of single DNA molecules.” Proceedings of the National Academy of Sciences 94.12 (1997): 6185-6190.
[3] Kowalczyk, Stefan W., et al. “Slowing down DNA translocation through a nanopore in lithium chloride.” Nano letters 12.2 (2012): 1038-1044.