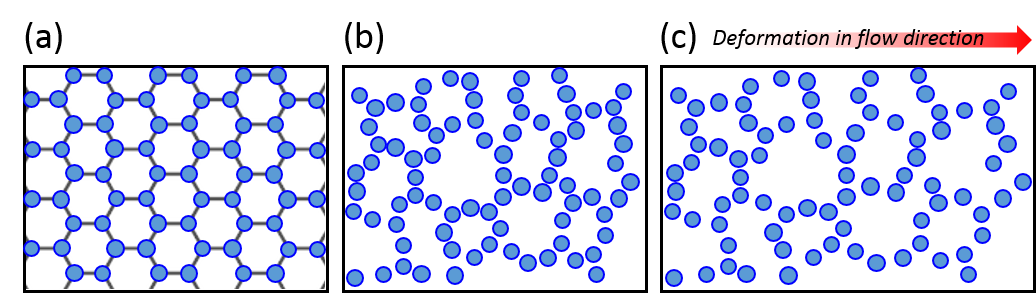Original paper: Rheology of Entangled Active Polymer-Like T. Tubifex Worms (arXiv here)
If you speak to a soft matter physicist these days, within a few minutes the term “active matter” is bound to come up. A material is considered “active” when it burns energy to produce work, just like all sorts of molecular motors, proteins, and enzymes do inside your body. In this study, the scientists are focusing specifically on active polymers. These are long molecules which can burn energy to do physical work. Much of biological active matter is in the form of polymers (DNA or actin-myosin systems for example), and understanding them better would give direct insight into biophysics of all kinds. But polymers are microscopic objects with complex interactions, making them difficult to manipulate directly. To make matters worse, physicists have yet to fundamentally understand the behaviors of active materials, since they do not fit into our existing theories of so-called “passive” systems. In this study, Deblais and colleagues decided to entirely circumvent this problem by working with a much larger and easier-to-study system that behaves similarly to a polymer solution: a mixture of squirming worms in water.
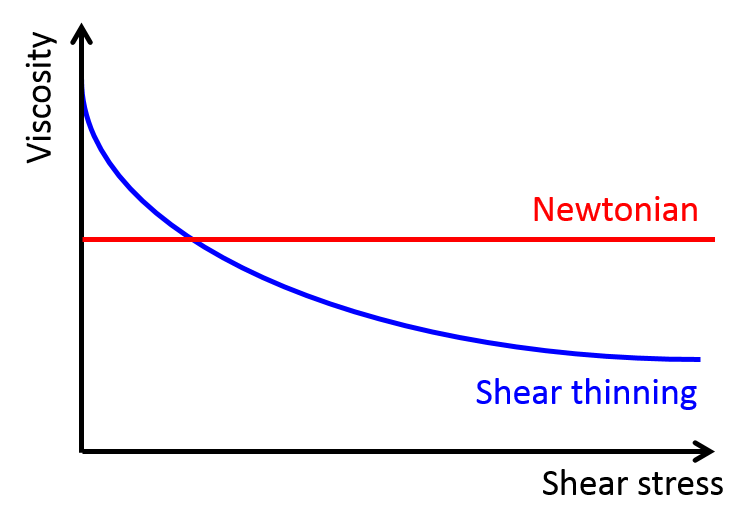
The researchers focused on the viscous properties of this living material, which behaves somewhat like a fluid. Viscosity is a measure of a fluid’s resistance to gradients in the flow. Polymer fluids are highly viscous because the long molecules in a polymeric liquid get tangled up in one another. Physical descriptions of most fluids assume that viscosity is a constant (so called Newtonian fluids), but many materials exhibit what is called shear thinning. This is when a fluid flows more easily as one applies an increasing shear force, that is, a force pulling the system apart. We encounter shear thinning at the dinner table all the time when struggling to pour ketchup, another polymeric fluid, out of a bottle. If the bottle is shaken fast enough, increasing the shear force applied, the ketchup flows smoothly like a liquid. In polymer systems (like xanthan gum in the ketchup) shear thinning happens when polymers are pulled apart fast enough that they tend to align together, which loosens the entanglements that held the system together before.
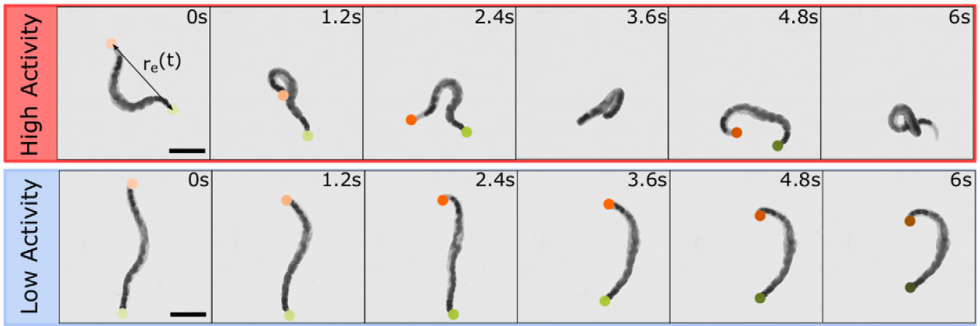
In this study, the researchers asked: how does shear thinning behavior change if the polymers in question were alive? To answer this question, they set out to measure the shear thinning properties of a mixture of worms at various levels of worm activity. Here, “worm activity” refers to how fast the worm is wriggling, which is calculated by measuring how quickly the distance between the two ends of a given worm changes. This leads to two logistical questions: how is the level of worm activity being modified, and how is the viscosity being measured?
Figure 1. This movie shows two worms, one in water (left) and one in a water + alcohol mixture (right). The worm on the right shows a decrease in activity when they are exposed to alcohol, which is one of the two ways the researchers modified worm activity in this study. Video taken from the original article.
The answer to the first question should be familiar to many humans. To make the worms less active, they were put into a solution containing water and a small amount of ethanol, the same type of alcohol found in beer, wine, and spirits. Once the worms were nice and drunk, the researchers noticed that they squirmed about more slowly, as shown in Figure 1. Thankfully, when the ethanol was removed, the worms returned to their previous level of activity! To make sure the alcohol wasn’t doing anything funny to the worms, they found a second way to reduce the activity — by reducing the temperature of the worm solution. Colder temperatures made for more chilled out worms, no pun intended.
Figure 2. This movie shows the functioning of the rheometer. The worms are placed inside a chamber between two plates. The top plate rotates with respect to the bottom plate, and the response of the material is measured. Video taken from the original article.
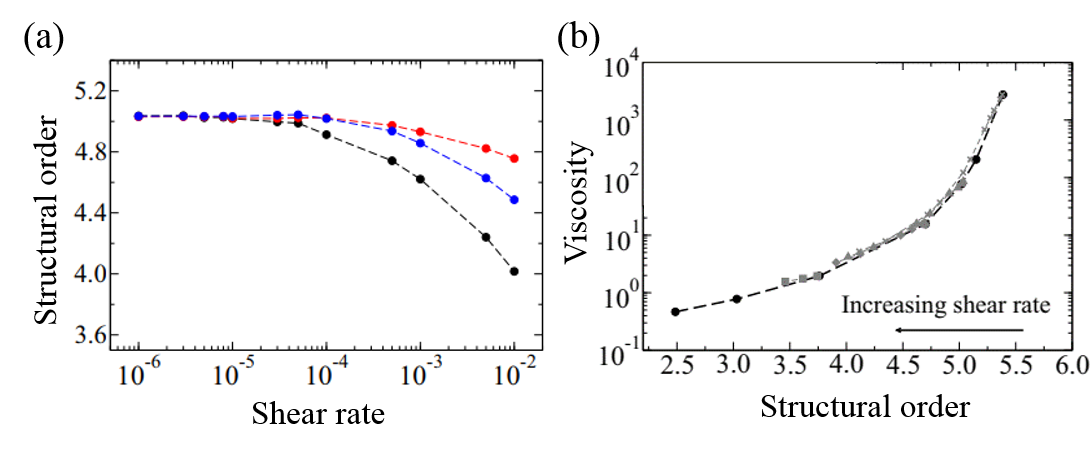
The researchers used a device called a parallel-plate rheometer to understand the shear thinning behavior of this living polymer system. As seen in Figure 2, a parallel-plate rheometer sandwiches a sample in between two flat plates and viscosity is measured by determining how much force is necessary to rotate the top plate, effectively pushing the material by twisting its surface. The viscosity of the worm mixtures was first determined at three different temperatures, and for worms drunk on ethanol. The results were surprising! The rheological behaviour of the low-activity worm mixtures matched with theories of polymer shear thinning quite well. It seems the worms have the same alignment properties as passive polymer solutions under shear!
So what happens when the worms are sober, more active, and wriggling around? They saw that the required twisting rate needed to thin the mixture decreased. In this case, the worm activity allowed for easier and quicker rearrangement while the mixture was pulled apart by the rheometer’s twisting motion. One can imagine that instead of needing to pull all the worms to the point of alignment, it may have been enough to nudge them in that direction and their wriggling did the rest. We can now imagine that the same thing might be true for non-living polymers: if a polymer material with shear thinning behavior is given an extra source of activity, then its thinning behavior may become more significant.
The lesson to be learned here is partly about worms, polymers, and the adverse effects of ethanol, but really this experiment is a testament to the power and generality of physical descriptions. This study teaches us about the possible behavior of an active polymer system with processes that are relevant on the scale of a few micrometers, by studying real life worms that you can see with the naked eye! In general, it is usually possible to find analog systems that have the desired properties for your study, but which are easier to manipulate. Physics then gives you the bridge between the system of interest and your simpler analog, allowing you to harness the power of interdisciplinary science to ask questions previously unanswerable.